Characterization of Trace Impurities in 3D-Printed Recycled ABS Materials
Home » Characterization of Trace Impurities in 3D-Printed Recycled ABS Materials
Traditional recycling processes can give rise to numerous deleterious effects on the molecular integrity of plastic materials. In the case of acrylonitrile-butadiene-styrene (ABS), these include polymer degradation, chain scission and unwanted cross-linking of the polymer chains.1 In addition, there is a notable loss of small molecules such as short chains or additives, which may volatilize during melt-processing of the materials.2 Furthermore, there may also be irreversible morphological changes, such as the formation of voids.3
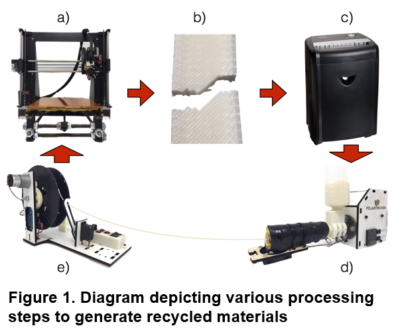
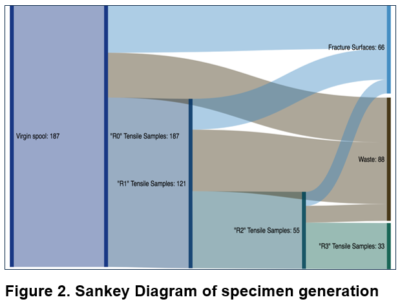
Herein we address these shortcomings using a novel recycling method that employs 3D printing. Our method involves fused deposition modeling (FMD), wherein a heated nozzle and motorized feed system are used to extrude a melted thermoplastic from a spool of filament. Deposited patterns of molten thermoplastic are continually laid onto a flat stage, moving up one layer at a time until the print is complete. The resulting material can then be shredded into a granulated material using a commercial paper shredder and then 3D printed into new filament as shown in Figure 1. Starting from virgin R0 plastic, we then repeated the process two more times to create 3× recycled materials (see Figure 2), which were characterized using a variety of analytical tools including primarily XRF, as well as FTIR, DSC and TGA.
XRF ANALYSIS
XRF (X-Ray Fluorescence) was performed using a Rigaku Primus II WDXRF with a rhodium X-ray source, vacuum atmosphere and an analysis area of 20 mm diameter. This analysis utilized a wavelength dispersive spectrometer (WDXRF) that is capable of detecting elements from atomic number (Z) 4 (beryllium) through atomic number 92 (uranium) at concentrations from the low parts per million (ppm) range up to 100%, by weight. Quantification was performed using the Fundamental Parameters (FP) standardless quantification software associated with the system. The fundamental parameters approach uses X-ray physics coupled with established sensitivity factors for pure elements.
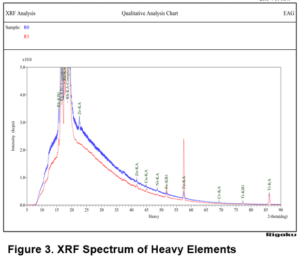
XRF analysis demonstrated an increase in the amount of titanium and iron during each processing step. This can be clearly observed in Table 1 and in Figure 3, which shows the growth of the Fe and Ti peaks. This result may be attributed to the wearing/shedding of steel components used for the processing and printing of the plastic.
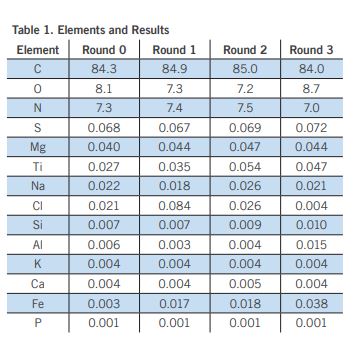
In addition, a slight increase is observed in the aluminum level from 0.006 wt% in R0 to 0.015 wt% in R3. Again, this could be attributed to the metal components used in the processing. The rest of the elements including Na, Si, K, Ca and P do not exhibit any significant changes. The carbon content remained between 84 and 85 wt%, while the oxygen content varied between 7.2 to 8.7 % and did not show any obvious trends. The nitrogen content varied between 7 to 7.5%. All other elements did not change significantly with each recycling step, suggesting that the process does not introduce a lot of defects or additional contaminants.
FTIR ANALYSIS
Fourier Transform Infrared (FTIR) spectroscopy was performed in transmission mode by removing a small portion of the ABS plastic and transferring to an infrared transmitting substrate. The samples were examined using a Thermo-Nicolet 6700 FTIR spectrometer equipped with a Continuum microscope. The analytical spot size was approximately 100 x 100 µm. OMNIC 8.0 software was used to perform data analysis.
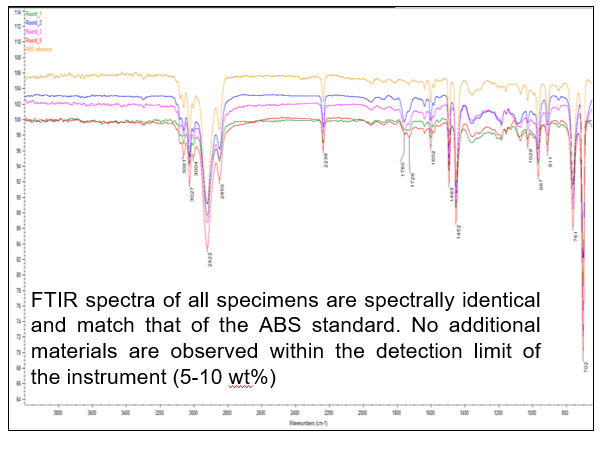
As shown in Figure 4, the FTIR spectra of all specimens are spectrally identical and match that of the ABS standard. No additional materials are observed within the detection limit of the instrument.
MOLECULAR WEIGHT AND THERMAL PROPERTIES
Polymer molecular weight was determined using GPC analysis on an Agilent 1100 Gel Permeation Chromatography system with two Resipore 300×7.5mm columns in series in THF. The instrument was equipped with an auto sampler and a Refractive Index Detector. The molecular weights were calibrated using polystyrene standards.
Thermal analysis was performed on a PerkinElmer DSC7 from 25 to 250 °C at 10 °C/min under a nitrogen atmosphere. TGA analysis was performed on a PerkinElmer TGA7 from 25 to 800 °C at 20 °C/min under a nitrogen atmosphere.
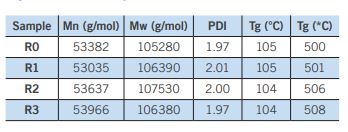
As shown in Table 2, no significant change in the molecular weight or PDI was observed in the recycled samples. Similarly, only a slight decrease in the glass transition temperature of the material, suggesting that the thermal properties have not substantially changed. The decomposition temperature of the polymer increased slightly with increasing R value, possibly due to the removal of low-level additives during the repeated recycling process.
FUTURE WORK
Additional experiments by XRD may help identify the iron and titania species detected by XRF. Furthermore, FTIR is not particularly sensitivem so TOF-SIMS and/or GCMS might be more useful in looking at low level organic compounds. Mechanical tests have been performed on these samples and are beyond the scope of this App Note.
REFERENCES
1Bai, X., Isaac, D. H., & Smith, K. (2007). Polymer Engineering and Science, 47, 120-130.
2Arnold, J. C., Alston, S., & Holder, A. (2009). Polymer Degradation and Stability, 94, 693-700
3Rahimi, M., Esfahanian, M., & Moradi, M. (2014). Journal of Materials Processing Technology, 214, 2359-2365.
Would you like to learn more about Characterization of Trace Impurities?
Contact us today for your impurities in 3D-printed recycled ABS material needs. Please complete the form below to have an EAG expert contact you.