Inductively Coupled Plasma Mass Spectrometry (ICP-MS)
Home » Our Techniques » Mass Spectrometry » ICP-MS
Inductively Coupled Plasma-Mass Spectrometry (ICP-MS) is one of the most important spectrometric techniques. It is a multi-elemental technique with extremely high sensitivity and large linear dynamic range, which allows simultaneous analysis of main components and ultra-trace elements. It is capable of analyzing elements from Li to U and can be applied to solutions and solids. Solid samples can be analyzed directly (by coupling the ICP-MS to an UV Laser Ablation system) or after dissolution or digestion using a combination of acids, heating and/or pressure. Typical digestion methods are: Microwave digestion (as this is a closed system, potentially volatile analyte species are preserved), Carius digestion (closed system), ashing (furnace), hot plate and parr bomb. The most suitable technique is used for each analysis and depends on the sample composition and characteristics. The resulting sample solution is then nebulized, after which the generated aerosol is transported into the core of an inductively coupled argon plasma, where temperatures of approximately 9000 K are attained. At such high temperatures, the nebulized solution is vaporized, and the analyte species are atomized and ionized. Ions generated in the high temperature argon plasma core are subsequently accelerated into a mass analyzer for both elemental and isotopic analysis. Intensity measurements are converted to elemental concentration by comparison with calibration standards.
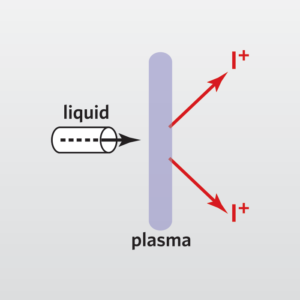
Method development (and/or validation) is commonly excised to establish the suitable protocols to analyze elements of interest for different matrices, with the acceptable precision, accuracy, detection limits, quantification limits and robustness. This technique is especially powerful for quantitative chemical analysis of high purity solids and liquids.
Ideal Uses of IC-MS
- Quantitative analysis of minor (μg/g in solids; mg/L in liquids) and trace (ng/g in solids; ng/L-μg/L in liquids) elements
- In solids, liquids and slurries
- On surface, in thin film and in bulk
- Leachables and extractables
- Purity certification, cleaning validation and contamination identification for a broad range of materials and processes of industrial significance for aerospace, automotive, defense, energy, mining, petroleum, pharmaceuticals and semiconductor industries, for example
- Ultra-pure water, cooling water, acids, organic solvents and lithograph stripping solutions
- High purity oxides and salts, including glass and high-performance ceramics
- High purity carbon materials, including graphite
- Semiconductor materials
- High purity metal targets and advanced alloys
- Organic polymers
- Drugs, food supplements, biomaterials and medical devices
- Other materials
- Supply chain efficiency support, ranging from raw materials/feedstock qualification, product safety, research and development, production control to failure analysis
- Medical device chemical characterization via ISO 10993:18
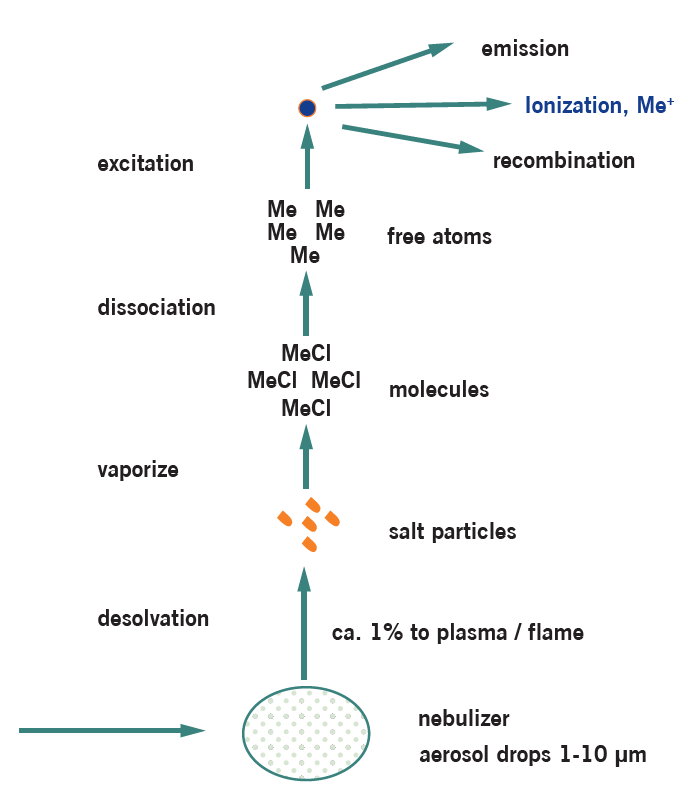
Strengths
- A wide range of elements (up to 70) can be measured in a single analysis
- The useful linear dynamic range is over several orders of magnitude, allowing simultaneous analysis of main components and trace elements
- High sensitivity, resulting in low detection limits
- The analysis can be automated, thus enhancing accuracy, precision and throughput
- Dynamic Reaction Cell (DRC), kinetic energy differentiation (KED), triple quad (TQ) and the High Resolution (HR) technique virtually eliminate polyatomic and isobaric interferences
Limitations
- Sample preparation is key and time-consuming
- For bulk analysis, the sample to be analyzed must be completely digested or dissolved prior to instrumental analysis
- Minimal risk of contamination during sample preparation
- Destructive
- Limited to <0.2% dissolved solids
- In mass spectral analysis, common matrix elements and other molecular polyatomic species can interfere with the measurement of some analytes
- Multiple-charged ions or isobaric interferences can create challenges in quantitation
- Carbon, nitrogen, hydrogen and oxygen cannot be determined using this technique
IC-MS Technical Specifications
- Signal Detected: Positive ions of (stable) isotopes (MS)
- Sample size requirements: 0.01-10 gram of solids; 1-100 mL of liquids
- Elements detected: Up to 70 elements (from Li to U)
- Typical detection limits: ng/g to μg/g in solids; ng/L to μg/L in liquids
- Spatially resolved analysis: Only in combination with Laser Ablation
Would you like to learn more about using ICP-MS?
Contact us today for your Inductively Coupled Plasma Mass Spectrometry needs. Please complete the form below to have an EAG expert contact you.