Part 18: Chemical Characterization of Materials
Part 18: Chemical characterization of materials, part of ISO 10993 pertains to evaluating biocompatibility and safety of medical devices.
For medical device manufacturers, Eurofins EAG Laboratories offers unmatched capabilities and analytical expertise, a unique range of in-house techniques and instruments, and fast, flexible problem solving.
EAG Laboratories applies vast experience with the materials that comprise medical devices to assist our clients in performing the necessary chemical characterization studies per ISO 10993-18 and the EU MDR. We are able to identify bioavailable chemicals released from a product through simulated use, exaggerated, or exhaustive extractions.
From R&D design screening to lot release and regulatory submission, manufacturers and suppliers are looking for in vitro testing that provides materials expertise with quick turnaround. As materials experts, we offer a greater understanding of why a certain material failed an assay. High throughput for in vitro testing is important. But when a material fails an assay, understanding why is crucial.
EAG has developed expertise in identifying and characterizing particles of an unknown origin with individual and combined compositional analysis techniques in conjunction with expert data interpretation. We have developed a library of spectra to accelerate particle identification investigations.
As ISO 10993-19 continues to evolve, we have pioneered the utilization of analytical techniques to meet industry-expected needs. EAG Laboratories uses over 30 different surface characterization methods to provide answers to our customers throughout the medical device life cycle.
EAG performs nickel leaching testing per ASTM F3306-19 Standard Test Method for Ion Release Evaluation of Medical Implants.
EAG supports cleaning validations per ISO 19227 utilizing a variety of target specific and non-target specific techniques. Our analysis focuses on the residue remaining on the devices including:
Our engineering services provide comprehensive support of the advanced electronics used in medical devices, including chip-level ATE (automated test equipment) development, reliability and qualification testing (e.g. burn-in, temp cycling, electrostatic discharge (ESD) testing, etc.), circuit edit/debug, and root cause failure analysis using advanced investigative techniques like CT/3D x-ray, Laser Signal Injection Microscopy, and Dual Beam Focused Ion Beam (DB FIB) analysis.
As a leader in the field of semiconductor and electronics testing and failure analysis, our engineering teams have decades of industry experience and expertise with a comprehensive array of cutting-edge equipment to support a wide range of engineering needs.
We also have extensive battery characterization capabilities, including battery cycling, environmental testing, and full teardown services.
EAG Laboratories scientists are experts in medical device surface analysis, including the factors that affect biocompatibility, corrosion-resistance, crack-resistance, adhesion and bonding ability. Our laboratories investigate and understand the issues associated with surface modifications, including how surfaces may change and degrade with use and time, and also the effects of different surface finishes, such as electropolishing.
Download the Brochure Today!
Contact us today for your medical device testing needs. Please complete the form below to have an EAG expert contact you.

Part 18: Chemical characterization of materials, part of ISO 10993 pertains to evaluating biocompatibility and safety of medical devices.
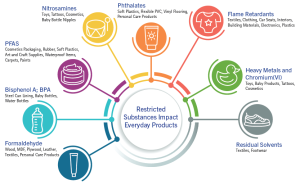
As the list of restricted substances grows, testing demands customized methods to identify issues early. This is a complex issue that requires a strategic approach.
Catheter and balloon analysis for medical device development support includes deformulation, contaminant identification and failure analysis.
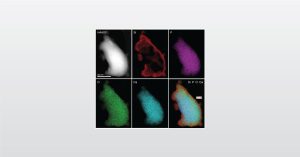
In this webinar we introduce Characterization of Bioceramics for Surgical Implants using analytical tools for full qualification
To enable certain features and improve your experience with us, this site stores cookies on your computer. Please click Continue to provide your authorization and permanently remove this message.
To find out more, please see our privacy policy.