X-ray Diffraction (XRD) Services
Home » X-ray Diffraction (XRD) Services
X-ray Diffraction (XRD) is a powerful nondestructive technique for characterizing crystalline materials. While most other analytical techniques provide elemental or molecular information from a sample, XRD is unique in providing a wide variety of information on structures, crystalline phases, preferred crystal orientations (texture), and other structural parameters such as crystallite size, percent crystallinity, strain, stress, and crystal defects.
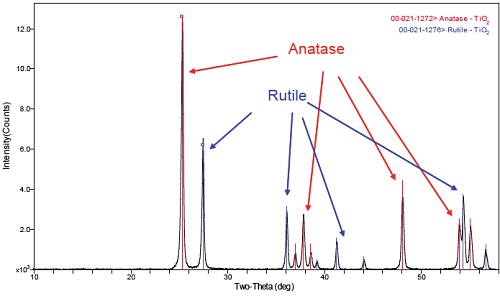
XRD has a very wide range of applications, across many sample types and materials. Please see our XRD application notes for more specific examples. A typical XRD application is phase-ID. Shown below is the diffraction pattern from a TiO2 sample.
The XRD results in figure 1 show that the sample contains both the Rutile (31.4%) and Anatase (68.6%) phases of TiO2. Compositional results would (ideally) show a 2:1 ratio of O:Ti, but these different phases of TiO2 have different physical and electronic properties so knowing which phase is present can be crucial.
PRINCIPLES
In XRD analysis a sample is exposed to a collimated X-ray beam of specific known wavelength. If the material is crystalline, it possesses a three dimensional ordering or “structure” with repeat units of atomic arrangement (unit cells). X-rays are diffracted by the repeating lattice spacings of crystalline materials, whereas they are simply scattered by amorphous materials. X-ray diffraction occurs at specific angles (2θ) with respect to the lattice spacings defined by Bragg’s Law:
nλ = 2d sinθ
where n is an integer denoting the order of the reflection, λ is the X-ray wavelength, d is the lattice spacing of the crystal planes of interest, and θ is the diffraction angle. Any change or difference in lattice spacing results in a corresponding shift in the diffraction lines. Consequently, the X-ray diffraction pattern is the fingerprint of periodic atomic arrangements in a given material. Diffraction patterns can be checked against large libraries of patterns from known materials in order to identify/quantify the phases present in a sample.
COMMON APPLICATIONS
While most other analytical techniques tend to provide elemental or molecular information, XRD provides crystalline information, resulting in many unique applications including:
- Identification/quantification of crystalline phase
- Measurement of average crystallite size, strain, or micro-strain effects
- Percent crystallinity versus amorphous component
- Texture (orientation): preferred orientation of crystallites
- Determination of lattice parameters to quantify alloy content
- Residual Stress – lattice compression or expansion High Resolution XRD (HRXRD) – Epitaxial layer composition, strain in/out of plane
- SAXS – Small Angle X-ray Scattering for agglomerated particle size and spacing
- High Temperature X-ray Diffraction – in-situ phase change measurement
STRENGTHS
- Provides a wealth of unique information about crystalline materials
- Sample the bulk of material (50µm-1cm depth), or near surface only (50-1000nm) using Grazing Incidence-XRD
- Able to uniquely identify specific crystalline phases
- Can measure lattice spacing
- Can analyze full wafers up to 300mm
- Minimal or no sample preparation requirements
- Non destructive, using ambient conditions for all analyses
LIMITATIONS
- Typical detection limit ~1%
- Cannot identify amorphous components
- Limited information with respect to depth (no depth profile, but Grazing Incidence-XRD can be used to increase sensitivity for thin layers)
- Smallest spot size is ~30µm
TECHNIQUE COMPARISONS
XRD is a good complement to bulk analysis techniques such as Glow Discharge Mass Spectrometry, Inductively Coupled Plasma-Optical Emission Spectroscopy (or Mass Spectrometry), and X-ray Fluorescence. While these other techniques provide compositional information, XRD provides crystalline information helping to provide a more complete characterization of the material. It should be noted that by itself XRD cannot provide unequivocal compositional information because it is possible that a sample may have an amorphous component that cannot be identified by XRD.
XRD can be quite powerful when combined with other techniques used for thin film analysis, such as Rutherford Backscattering Spectroscopy, Auger and X-ray Photoelectron Spectroscopy. XRD can provide phase, orientation, and other crystalline information to complement the compositional results from RBS, Auger, or XPS. XRD tends to probe much deeper than these techniques, providing more bulk information unless grazing incidence (GI-XRD) is used to enhance the surface sensitivity of the technique.
XRD AT EAG
EAG has five XRD systems located within our network of labs. Three of our instruments are equipped with optical modules that can be easily exchanged depending on the analysis requirement. This allows us to provide high quality analysis of powders, coatings, thin films, slurries, fabricated parts, or even high-resolution analysis of epitaxial films. Two systems have microbeam X-ray sources, allowing for the analysis of very small samples or specific locations. These tools also have area detectors allowing for complete texture analyses.
Would you like to learn more about X-ray Diffraction (XRD) Services?
Contact us today for your X-ray Diffraction (XRD) Service needs. Please complete the form below to have an EAG expert contact you.