Cause and Location of Delamination of Multi-layer Metal Foil by XPS
Home » Cause and Location of Delamination of Multi-layer Metal Foil by XPS
Determining the locus of multilayer laminate failure is often the first step in understanding the cause of failure. In many cases a fraction of a monolayer of contamination is enough to weaken a bond to the point of failure. In this example we examine mating faces from a failed Cu-laminate (see the figure below). X-ray photoelectron spectroscopy (XPS) is used because of its surface sensitivity and ability to quantitatively identify not only the elements present but also determine their chemical or oxidation states.
As expected, copper was detected on both sides of the failed interface. Significant quantities of Ni°, Ni3+, Mo°, Mo4+, Mo6+ and PO43- were detected on both sides of the failed interface by XPS. Carbon was also detected, but likely came from adsorption of organic species during air exposure prior to analysis.
To probe the thickness of the Ni, Mo and P containing layers, the exposed surfaces were depth profiled using an argon ion beam. The resulting profiles reveal a roughly 10Å thick phosphate layer on both sides of the failed interface. The P concentration is relatively low (2.5%); however, it was determined to be present as PO43-, meaning the total amount of phosphate at the interface is >5X the P concentration. The cation associated with the phosphate was likely either Cu, Mo or possibly H (hydrogen is not detected by XPS). Beneath the phosphate layer on the Carrier side was a ~100Å thick Ni2Mo layer. A small amount of Ni was detected on the mating foil surface.
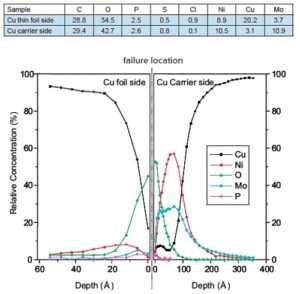
XPS was used to determine that adhesive failure occurred at a Ni2Mo-Cu interface as a result of a high concentration of phosphate.
Would you like to learn more about Multi-layer Metal Foil by XPS analysis?
Contact us today for your delamination of multi-layer metal foil needs. Please complete the form below to have an EAG expert contact you.