IGA Fractional O/N/H Analyses of Powder Feedstocks for Additive Manufacturing
Home » IGA Fractional O/N/H Analyses of Powder Feedstocks for Additive Manufacturing
Additive manufacturing calls for powder feedstock of metals, alloys and ceramics, with particle size typically in 100s nm – 10s μm range. This leads to orders-of-magnitude increase in surface area from the bulk materials. Driven by the increase in surface free energy (i.e., thermodynamic favorable) and the decrease in diffusion length (i.e., kinetic favorable), the surface chemistry of powders becomes equally important to, if not greater than, the bulk chemistry. Of particular interest is to understand and control the O, N and H chemistries of powders, which are prone to change depending on the material type, the powder manufacturing technique, working atmosphere, packaging/handling, etc.
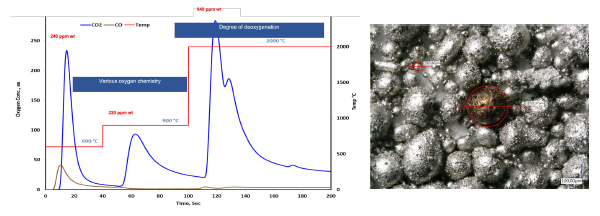
Interstitial Gas Analyses (IGA) is the standard method to determine bulk content of O, N and H in inorganic materials, such as metals, alloys and ceramics, via carbo-reductive inert gas fusion, or hot extraction-induced dehydrogenation, followed by infrared or thermal conductivity detection. Taking advantage of the temperature-programming capability of LECO® ONH836 instrument, herein we wish to demonstrate that by analyzing samples in different temperature modes (flash heating, ramping and stepped heating), it is possible to quantitatively speciate the O, N and H chemistries in powders, such as surface and interface oxygen, oxide precipitates, interstitial oxygen, surface -OH or physiosorbed moisture, from % levels to ppm levels.
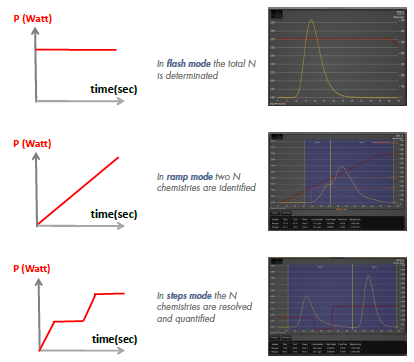
IGA fractional analysis gives:
- Information on various oxygen chemistries
- Surface oxygen/bulk oxygen ratio
- Quantitative and precise results
Would you like to learn more about Analyses of Powder Feedstocks?
Contact us today for your analysis of powder feedstocks. Please complete the form below to have an EAG expert contact you.