Zeta Potential Analysis
Home » Our Techniques » Zeta Potential
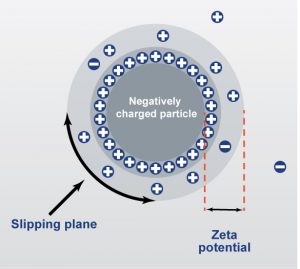
Zeta potential analysis is a scientific technique used to measure the electrical potential at the slipping plane of a particle in a liquid suspension. This measurement is a key indicator of colloidal stability, helping researchers predict whether particles will remain dispersed or clump together (agglomerate) over time.
When a charged particle is in a liquid, it attracts a thin layer of oppositely charged ions that move with it. The boundary of this moving layer is called the slipping plane, and the voltage at this point is the zeta potential.
Ideal Uses of Zeta Potential Analysis
The ideal use of Zeta Potential Analysis is to determine the surface charge and predict the long-term stability of colloids, emulsions, and suspensions, with higher absolute values (typically mV) indicating greater stability against aggregation. It is primarily used to optimize formulations in pharmaceuticals, paints, coatings, and water treatment.
Key applications and benefits include:
- Stability Optimization: Assessing, predicting, and optimizing the stability of emulsions and suspensions to prevent settling, flocculation, or phase separation.
- Formulation Development: Rapidly assessing new formulations in industries like food, cosmetics, and paints to ensure desired shelf-life and consistency.
- Surface Charge Characterization: Measuring the electrostatic repulsion or attraction between nano-sized particles to understand particle interaction.
- Isoelectric Point Determination: Identifying the pH at which a material has zero net charge (isoelectric point) to control stability or induce aggregation, which is critical for water treatment and material science.
- Additive Efficiency: Determining the effectiveness of surfactants or dispersants in reducing aggregation.
Strengths
- Predicts Dispersion Stability
- Rapid Formulation Optimization
- Broad Applicability
- Versatile Measurement Techniques
- Surface Charge Analysis
- Small Sample Requirements
Limitations
- High particle concentrations lead to multiple scattering, while high salt concentrations (high conductivity) interfere with electrical measurements and can cause electrode polarization.
- The technique struggles with polydisperse samples (yielding averaged values), non-spherical particles, and samples that sediment quickly.
- Small variations in pH, temperature, or solvent viscosity can significantly alter results.
- Measurements require careful handling of electro-osmotic flow (using the stationary plane) to avoid errors, and improper sonication can change particle characteristics.
- While ideal for simple colloids, accurately determining the zeta potential of complex, non-particle surfaces (like membranes or fibers) or in complex liquids is difficult.
Zeta Potential Analysis Technical Specifications
- Range: 4.0 nm – 100 μm
- Sample Volume: Minimum 1.0 mL
- Light Source: 40 mW 658 nm Laser
Would you like to learn more about Zeta Potential Analysis?
Contact us today for your Zeta Potential needs. Please complete the form below to have an EAG expert contact you.