
ESD Webinar
In this webinar we introduce electrostatic discharge (ESD) testing which is one of the failure mechanisms for integrated circuit parts
Home » Extraction Expertise for Medical Device Chemical Characterization
Extractable studies are performed on medical devices to evaluate potential chemical release from a medical device during clinical use.
The primary guidance for medical device extractable studies is ISO 10993-18 “Chemical characterization of medical device materials within a risk management process”. In this international standard, a number of extraction study approaches are laid out, which are applied to medical devices based on the intended device use.
EAG offers advanced expertise and a risk-based approach for medical device extractable study design and execution, including the latest FDA expectations.

ISO 10993-18 provides guidance for the type of extraction study necessary for chemical characterization, based on device contact.


In this webinar we introduce electrostatic discharge (ESD) testing which is one of the failure mechanisms for integrated circuit parts

Demand for compound semiconductors has rapidly grown recently. During this live event we answered questions surrounding the Materials Characterization of GaN HEMT power transistors.
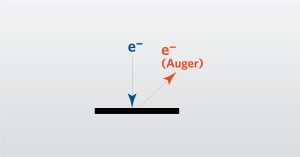
This webinar is an introduction to Auger Electron Spectroscopy. It will include the principles of the technique with examples.

There is still so much to learn about batteries, including challenges such as energy density, cycle life, fast charge, and safety. In this blog, we’ll be focusing on energy density.
To enable certain features and improve your experience with us, this site stores cookies on your computer. Please click Continue to provide your authorization and permanently remove this message.
To find out more, please see our privacy policy.