Root Cause Determination of Adhesion Failure in Medical Device Packaging
Home » Root Cause Determination of Adhesion Failure in Medical Device Packaging
A medical device sterile package experienced adhesive failure at a polyethylene-ethylene acrylic acid (co-polymer) heat seal to polyethylene. If undetected, these types of failures can lead to loss of sterility and result in serious harm to patients. Adhesion failures are frequently chemical in nature and can be caused by very low levels of surface contaminants. The two most common analytical techniques to investigate surface chemistry on polymers are X-ray Photoelectron Spectroscopy (XPS) and Time-of-Flight Secondary Ion Mass Spectrometry (TOF-SIMS).
XPS is a quantitative technique able to both detect elements and obtain information regarding nearest neighbor chemical bonding. The sampling depth of XPS is ~5-10 nm. TOF-SIMS provides high mass resolution maps of elements and molecules in the top 1 nm of a sample. TOF can be invaluable for identifying organic compounds on surfaces.
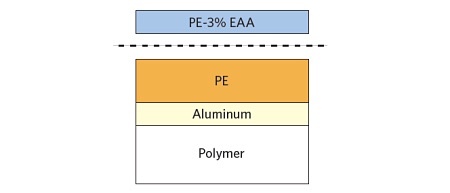
In investigations of adhesion failures it is desirable to examine mating sides since the contaminants may preferentially transfer to one surface. This approach can also help confirm the locus of failure which is not always straightforward in multilayer laminates containing transparent materials. A schematic of the laminate is shown with the failure path.
RESULTS AND DISCUSSION
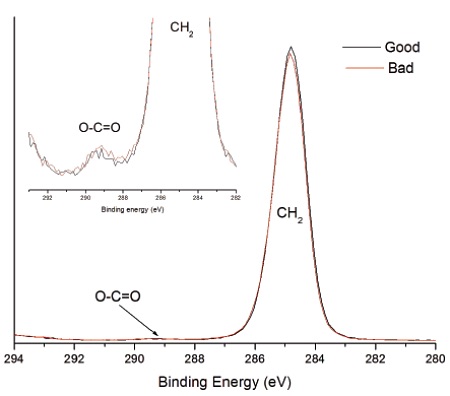
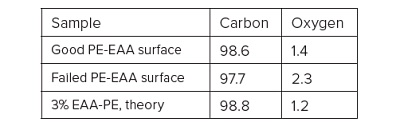
Ethylene acrylic acid (EAA) was added at 3% to improve the adhesion and lower the crystallinity of the polyethylene (PE). Good and bad heat seal surfaces were examined using XPS and TOF-SIMS. XPS found only carbon and oxygen on the surfaces, although the failed heat seal surface contained ~60% more oxygen than the good surface (Table 1). The high resolution carbon spectra are overlaid in Figure 2. The inset confirms the presence of a –CH2 and a small amount of O-C=O on both surfaces. The O-C=O is expected from the acrylic acid. If we assume that all the oxygen comes from EAA, the Good heat seal contains the expected 3% amount of EAA, but the Bad heat seal contains closer to 5% EAA. There is a second possibility to explain the excess oxygen on the failed surface: there could be a contaminant that also contains -CH2 and O-C=O species. To sort out these two possibilities, we turned to TOF-SIMS with its ability to detect and identify organic molecules on surfaces.
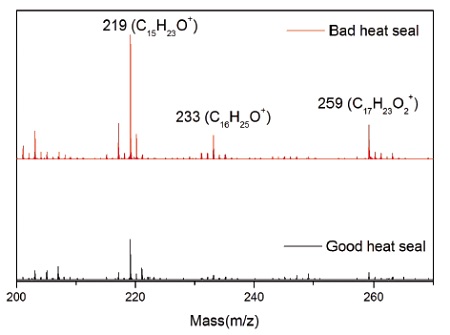
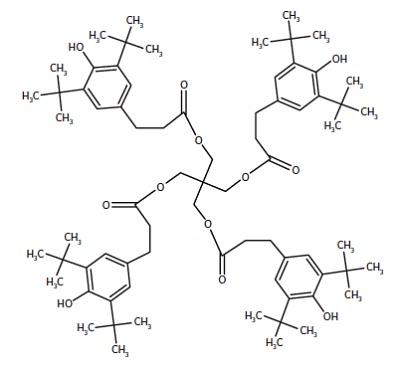
Both surfaces contained ions indicative of PE (C2H3, C3H5, C4H7, etc.) and EAA (CH3O, C2H5O, etc.). However, the Bad heat seal also contained intense peaks characteristic of a hydroxyhydrocinnamate compound (Figure 3). Such compounds are common antioxidants under the Irganox® brand. Figure 3 shows the molecule for Irganox® 1010, one of the common hydroxyhydrocinnamate compounds. Weak hydroxyhydrocinnamate peaks were observed on the Good heat seal surface at roughly 1/3 the intensity.
SUMMARY
XPS was able to identify an alkyl carbon and a weak O-C=O band on both good and bad surfaces. Quantitative results suggested either the presence of elevated EAA levels on the bad surface or possibly a acid or ester-containing contaminant. The molecular specificity of TOF-SIMS conclusively showed that the excess oxygen did not come from EAA, but from elevated levels of an antioxidant on the heat seal surface. It was concluded that this was the root cause of the adhesion failure.
Would you like to learn more about Adhesion Failure in Medical Device Packaging?
Contact us today for your adhesion failure in medical device packaging needs. Please complete the form below to have an EAG expert contact you.