Chemical analysis of glass defects
Home » Chemical analysis of glass defects
The chemical composition of all kinds of glass defects can be determined by SEM combined with EDS, XRF, Auger, and RGA.
Scanning Electron Microscopy
SEM is an imaging technique, making use of emission of electrons from a surface when irradiated by a high energetic electron beam. The information in the images is based on either topography or composition. The X-rays that are emitted as a result of the electron interaction with the sample provide quantitative information on the local chemical composition. The chemical composition together with the optical and microscopic observations, could give a good impression of the root cause of the corresponding glass defect.

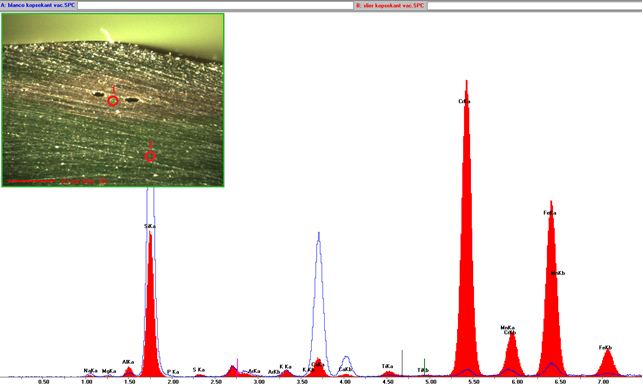
(µ-) X-ray fluorescence
(µ-) X-ray fluorescence is a well-established analytical technique for the determination of the elemental composition of solid materials. Its speed, reliability and accuracy make it extremely useful for process development/control and process optimization and therewith for the chemical analysis of foreign particles embedded in glass. Irradiation of a glass defect by an X-ray beam will result in the emission of element specific secondary X-rays. The composition of the embedded material can be verified by comparison of the spectrum of the glass defect with the surrounding glass matrix. Based on this information the origin can be determined.
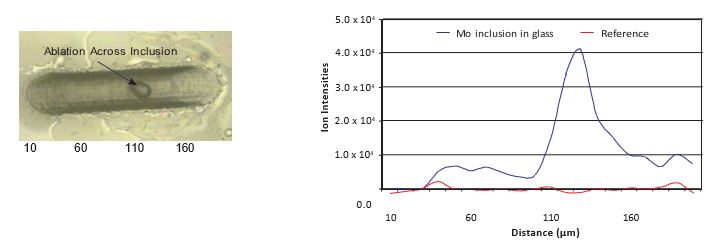
Laser Ablation-Inductively Coupled Plasma-Mass Spectrometry
Sometimes defects can be due to impurities present at low concentration levels (ng g-1 or μg g-1). In that case Laser Ablation-Inductively Coupled Plasma-Mass Spectrometry (LA-ICP-MS) is suitable to determine the composition (from main components to trace elements) and therewith the origin of the glass defect. By using an UV-laser, a small amount of material is sampled. The composition is subsequently analyzed by means of ICP-MS. A major advantage of the technique is that the inclusion does not have to be present at the surface of the glass. The laser can drill in the glass while determining the composition in depth or lateral. No or limited sample preparation is required, which is an advantage in case small inclusions must be analyzed.
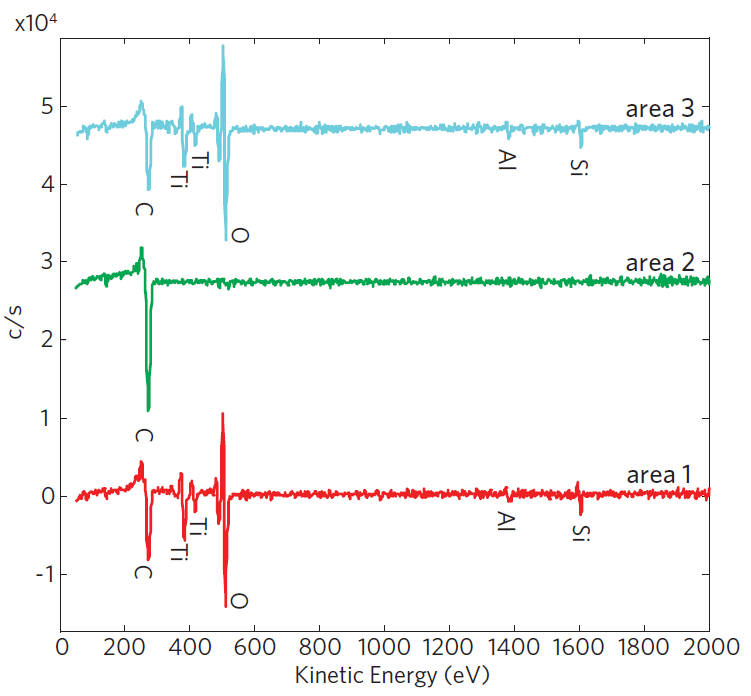
Auger Electron Spectroscopy
Surface particles, defects and discolorations can affect appearance. Pinholes may affect corrosion of silver layers. Auger Electron Spectroscopy (AES) is a surface analysis technique with shallow sampling depth (approximately 5 nm), which can be used to examine sub-micron features. It provides quantitative elemental and chemical state information and can be applied to examine small particles, stained areas and corrosion sites.
Auger can determine:
- Particle composition
- Discoloration composition
- Defect locations and residues
- Corrosion site products

Residual gas analysis (RGA), mass Spectrometry

Gas composition of inclusions (bubble)
In the production process of glass products, small gas inclusions can be created. Although some see it as art, these small gas inclusions can be highly undesirable from a cosmetic point of view or even prevent the product to meet its specifications. The composition of small gas inclusions can be analyzed using a mass spectrometer (MS). The results can help to trace and solve problem sources in the manufacturing process. MS is a well-established analytical technique used for the determination of gas compositions and pressure. Its speed, reliability and accuracy make it very useful for process development, control and process optimization and root cause analysis. Reliable measurements can be performed on gas inclusions with a diameter down to 80-100 µm.
Analysis of gas inclusions
First, the bubbles in the glass are located and inspected. Subsequently, the part containing the bubble is cut out. The samples are prepared in such a way that during the breaking procedure the captured gas is expanded into the mass spectrometer measurement chamber. The gas is ionized, and its composition is measured with the mass spectrometer. Calculation with factors obtained from calibration gasses, reveal both concentration and partial pressures of individual components in the gas inclusion.
Typical gasses to be measured are: H2, He, CO2, N2, SO2, COS, CO, H2S, O2, Ar, Ne, Xe and H2O. On request, other type of gasses can be added depending on the furnace conditions.
Possible source to be identified:
- Electrolysis
- Melting / refining
- (Fast) melting / refining
- Refractory interaction
- Contamination
Would you like to learn more about Chemical analysis of glass defects?
Contact us today for your chemical analysis of glass defects needs. Please complete the form below to have an EAG expert contact you.