ICACC 2025
January 28-29, 2025
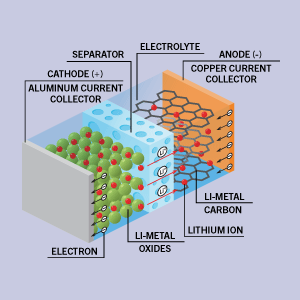
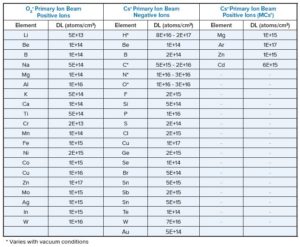
Eurofins EAG Laboratories will have a poster presentation and exhibit at ICACC 2025 in Daytona Beach, FL. MD&M West in Anaheim. Gideon Bartov, Senior Scientist with EAG, will present during Poster Session A on Tuesday, January 28th starting at 5pm.