
Effects of Surface Roughness
The roughness of a surface and how it interacts with surrounding materials and elements can have a significant impact on material technology and its functionality.
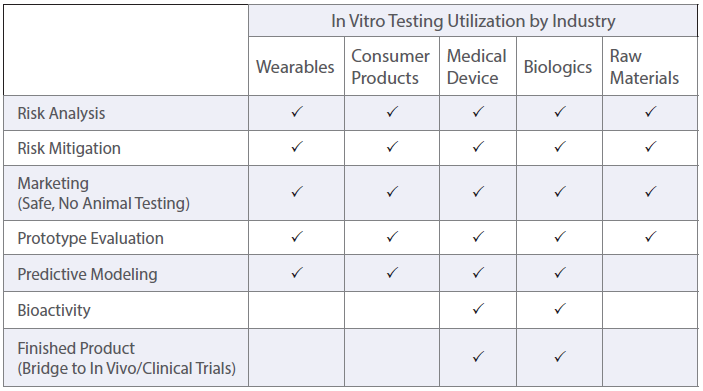
Home » Principles and Strengths of In Vitro Testing
In vitro is the Latin term for “in glass,” meaning that the testing is performed in a container that is outside of a living organism. This testing uses cell-based biological models instead of animals or humans. In vitro efforts help fulfil the FDA’s “3Rs approach” to replace, reduce, and/or refine animal testing.
Replace
Reduce
Refine
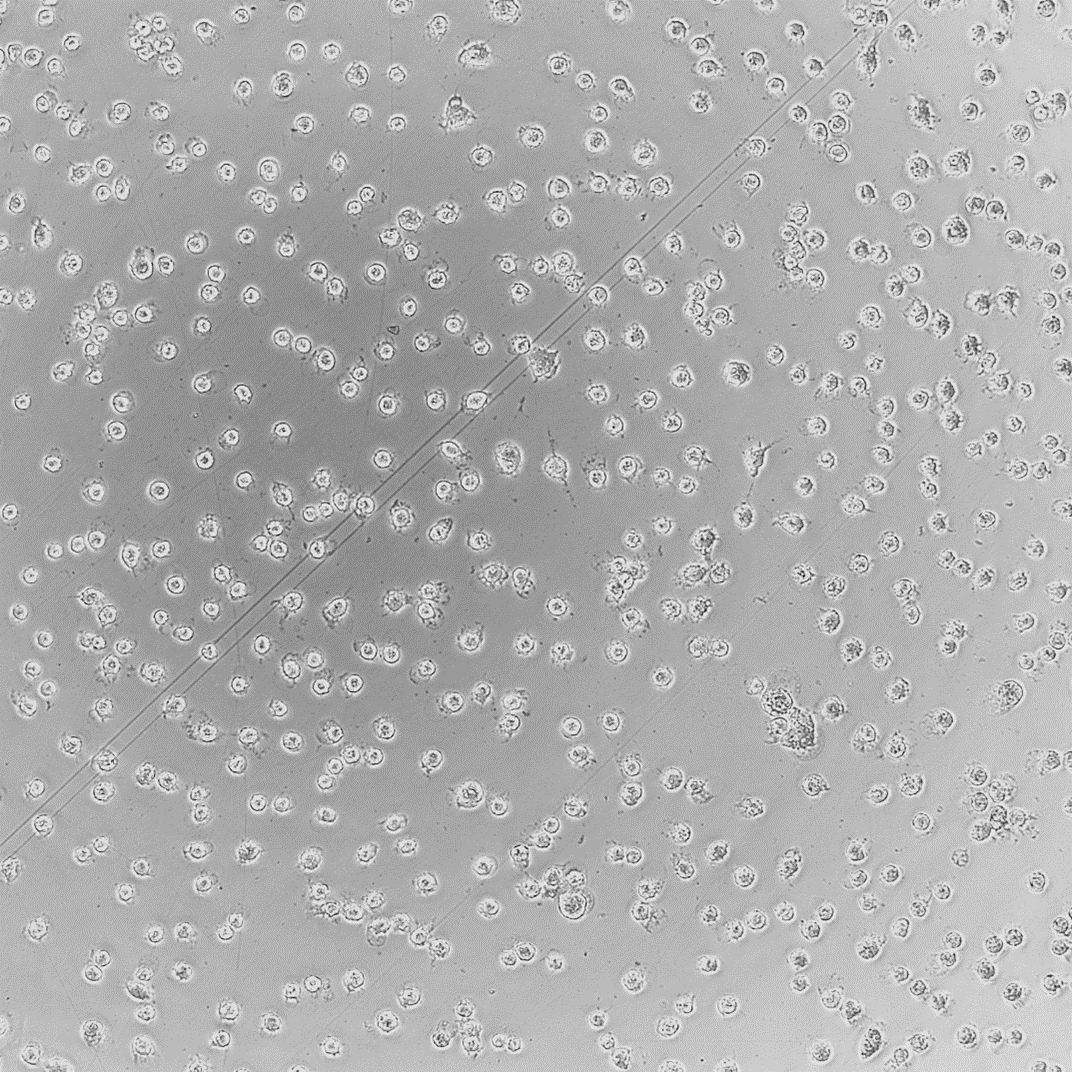
In Vitro model systems can be specified to the test sample’s intended use. 2D cell cultures are grown rapidly for high-throughput or multi-sample analysis with minimized variation between treatments. Cell cultures can also be made into 3D tissue models. These models contain differentiated cell layers to physiologically represent complex tissues and evaluate specific endpoints. Using specific geometries, in vitro cell culture models can be used to evaluate simple and complex biological responses.
To assess different biological endpoints, various forms of analytical methods may be implemented. Measurable responses of the in vitro models include cell death, growth inhibition, genetic alterations, changes in surface marker expression and altered metabolism.


The roughness of a surface and how it interacts with surrounding materials and elements can have a significant impact on material technology and its functionality.

Join us for an engaging virtual info session titled “Cultivating Talent & Community,” hosted by EAG Laboratories in collaboration with our parent company, Eurofins Scientific.
In this webinar we introduce the analytical approaches for solving food and beverage challenges including contamination.

Unwanted chemical impurities can be problematic. GDMS is a powerful full survey analysis tool for chemical purity evaluations.
To enable certain features and improve your experience with us, this site stores cookies on your computer. Please click Continue to provide your authorization and permanently remove this message.
To find out more, please see our privacy policy.