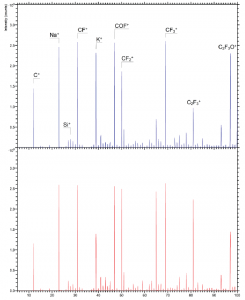
Chemical Characterization of Medical Devices How Creativity and Care Play a Role in Effective Study Design
Biocompatibility testing of medical devices per ISO 10993-18 is a resource intensive process. Careful front-end study design leverages efficiencies to reduce timelines and minimize the total number of devices
required for testing.